In sp hybridization, one 2s and one 2p orbitals of excited
carbon intermix to form two sp-hybrid orbitals in linear geometry. Each
sp hybrid orbital is occupied by one electron. The remaining pure 2p
orbitals ( for our convenience, let us say: 2py and 2pz) orient at right angles to the sp-hybrid orbitals. These are also occupied by one electron each.
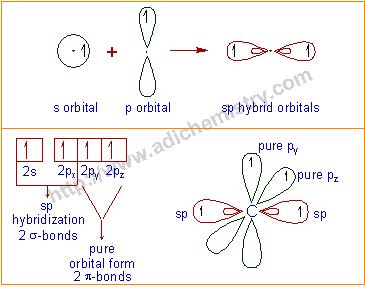 The two sp hybrid orbitals form 2 σ-bonds in linear geometry. Thus the bond angle will be about 180o. The remaining pure 'p' orbitals will form two π-bonds. Thus carbon again forms total four bonds i.e., two σ-bonds and two π-bonds.
The two sp hybrid orbitals form 2 σ-bonds in linear geometry. Thus the bond angle will be about 180o. The remaining pure 'p' orbitals will form two π-bonds. Thus carbon again forms total four bonds i.e., two σ-bonds and two π-bonds.
E.g., In acetylene molecule, C2H2, each carbon undergoes sp hybridization and forms one σ-bond with a hydrogen atom and one σ-bond with another carbon. The two carbon atoms also form two π-bonds with each other due to sidewise overlapping of pure p-orbitals. Thus a triple bond is formed between two carbon atoms in acetylene molecule.
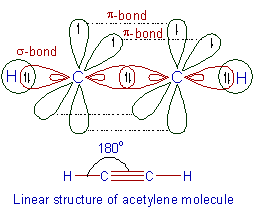 Note that whenever carbon atom undergoes sp hybridization, it forms 2 σ-bonds and 2 π-bonds. It may either form one triple bond as in case of acetylene or two double bonds e.g. allenes.
Note that whenever carbon atom undergoes sp hybridization, it forms 2 σ-bonds and 2 π-bonds. It may either form one triple bond as in case of acetylene or two double bonds e.g. allenes.
The following diagram summarizes the bonding pattern by carbon atom in different hybridizations.

E.g., In acetylene molecule, C2H2, each carbon undergoes sp hybridization and forms one σ-bond with a hydrogen atom and one σ-bond with another carbon. The two carbon atoms also form two π-bonds with each other due to sidewise overlapping of pure p-orbitals. Thus a triple bond is formed between two carbon atoms in acetylene molecule.

The following diagram summarizes the bonding pattern by carbon atom in different hybridizations.
No comments:
Post a Comment